FDA: 2023 Medical Device User Fees Announcement
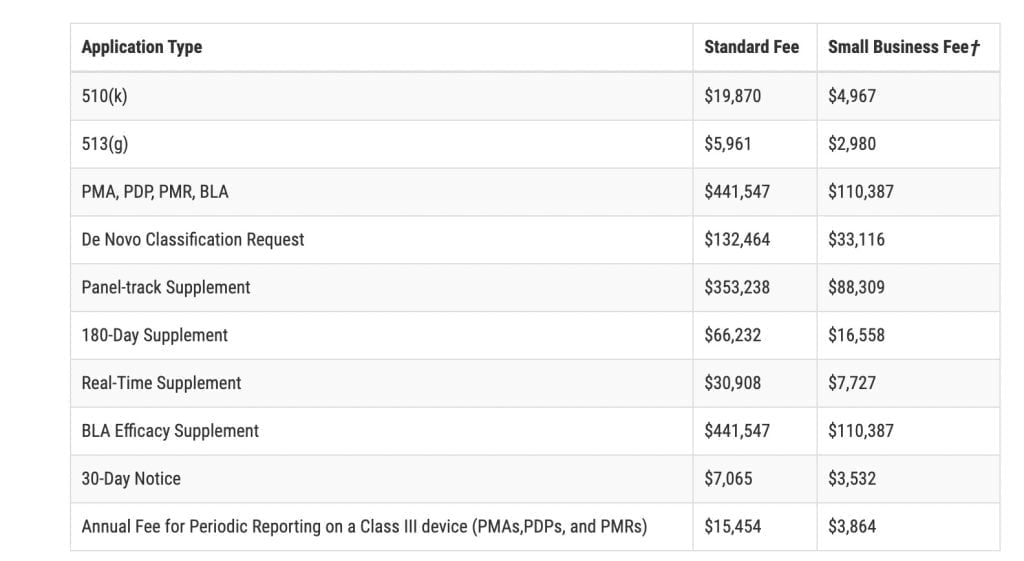
FDA: 2023 Medical Device User Fees Announcement Medical device user fees for the Fiscal Year 2023 are now available following an announcement from the U.S. Food and Drug Administration (FDA). The fiscal year runs from October 1, 2022 – September 30, 2023. All medical device facilities must pay the new fee amounts to keep their […]
FDA: 2023 Medical Device User Fees Announcement Read More »