REGULATORY NEWS AND RESOURCES
- All
- Canada Cosmetic Regulations
- Canada Dietary Supplement NHP NPN
- Dubai Cosmetic Regulations
- EU Cosmetic Regulations
- International Reuglatory News
- UK Cosmetic Regulations
- Uncategorized
- US FDA Dietary Supplements
- US FDA food regulations
- US FDA Medical Devices
- US FDA OTC regulations
- USA Cosmetic Regulations
All
- All
- Canada Cosmetic Regulations
- Canada Dietary Supplement NHP NPN
- Dubai Cosmetic Regulations
- EU Cosmetic Regulations
- International Reuglatory News
- UK Cosmetic Regulations
- Uncategorized
- US FDA Dietary Supplements
- US FDA food regulations
- US FDA Medical Devices
- US FDA OTC regulations
- USA Cosmetic Regulations
What is the Fragrance allergens update EU?
Pasquale Carvelli
August 6, 2023
Fragrance allergens update EU The European Commission issued a new regulation (EU) 2023/1545 on July 27, 2023, updating the list of fragrance allergens ...
Post-Market Surveillance for Cosmetics in Europe and the USA
Pasquale Carvelli
May 9, 2023
A Comprehensive Look at Post-Market Surveillance for Cosmetics in Europe and the USA Cosmetics have become an essential part of our daily lives, with ...
Update on UKCA Cosmetic Labeling and Marking Requirements
Pasquale Carvelli
May 4, 2023
Update on UKCA Cosmetic Labeling and Marking Requirements: Extended Transition Period Announced by UK Government On November 14th, 2022, the Government of the United Kingdom ...
FDA VCRP Discontinuation
Pasquale Carvelli
April 7, 2023
FDA Halts VCRP in Anticipation of Compulsory Cosmetics Registration The FDA’s latest cosmetic regulation update has shaken the beauty industry. According to the constituent update ...
14 US Agent Questions to Know to Meet Medical Device Regulations
Pasquale Carvelli
February 20, 2023
14 Key Questions About US Agents to Ensure Compliance with Medical Device Regulations Medical device regulations must be met if you want to sell your ...
Latest Medical Device User Fee Amendment (2023)
Pasquale Carvelli
February 5, 2023
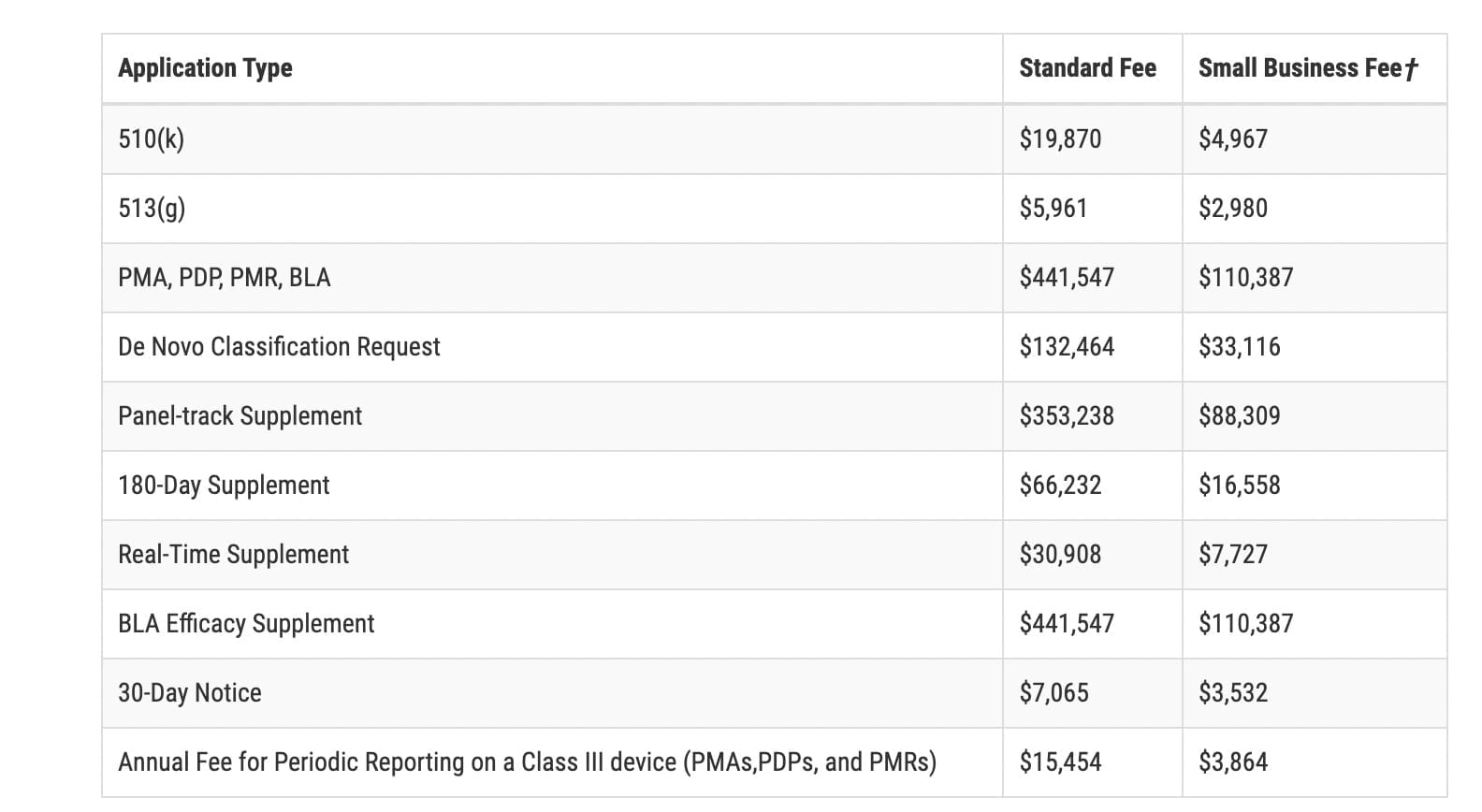
FDA: 2023 Medical Device User Fees Announcement Medical device user fees for the Fiscal Year 2023 are now available following an announcement from the U.S. ...
Key Developments in EU and UK Regulations: Early 2023 Updates
Pasquale Carvelli
January 21, 2023
Key Developments in EU and UK Regulations: Early 2023 Updates 1. Recent Developments in Chemical Regulations In recent years, there has been growing scrutiny on ...
FDA: 2023 Medical Device User Fees Announcement
Pasquale Carvelli
November 2, 2022
FDA: 2023 Medical Device User Fees Announcement Medical device user fees for the Fiscal Year 2023 are now available following an announcement from the U.S. ...
What’s the Difference Between Food and Dietary Supplement Labeling in the United States and Canada?
Pasquale Carvelli
July 14, 2022
What’s the Difference Between Food and Dietary Supplement Labeling in the United States and Canada? What’s the Difference Between Food and Supplement Labeling in ...